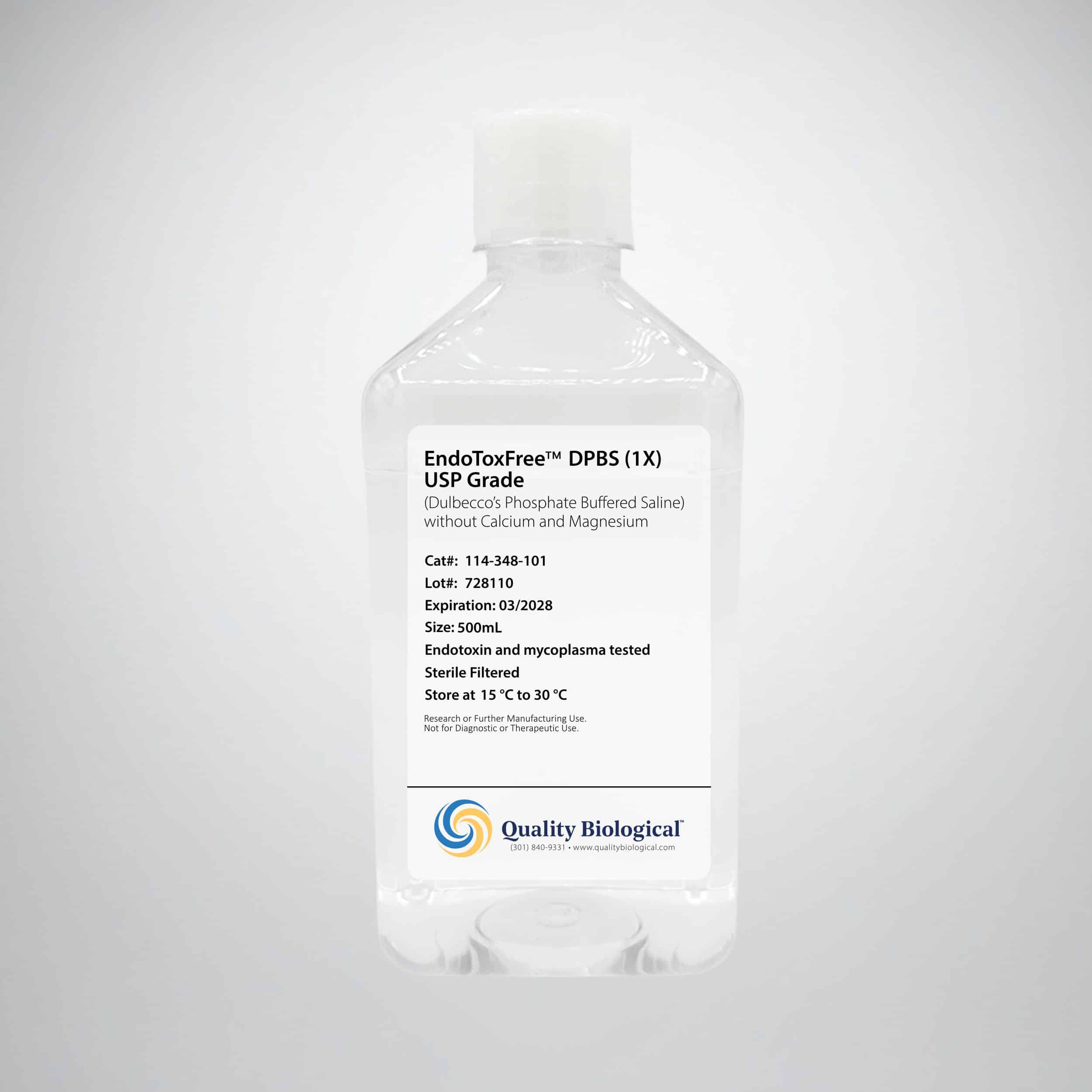
EndoToxFree™ DPBS (1X), USP Grade
(endotoxin levels <0.005 EU/mL)
Description
EndoToxFree™ DPBS (1X), is an ultrapure, USP Grade Dulbecco’s phosphate-buffered saline, formulated without calcium and magnesium. Manufactured under stringent conditions, ensuring endotoxin levels of <0.005 EU/mL, this buffer is specifically designed for sensitive cell culture and research applications where endotoxin contamination could compromise results.
KEY BENEFITS OF EndoToxFree™ DPBS (1X), USP Grade
- Reliable, reproducible results: Ultra-low endotoxin levels (<0.005 EU/mL) eliminate interference that can compromise your critical experiments and waste valuable time
- Seamless research-to-manufacturing transition: USP Grade quality ensures your protocols will scale smoothly from bench to production, reducing regulatory hurdles
- Enhanced experimental control: This calcium and magnesium-free formulation gives you precise control over cell behavior, preventing unwanted adhesion and ensuring clean enzymatic reactions
Research areas that benefit from Endotoxin Free Reagents
Reproducibility, sensitivity, and accuracy of your research can be directly impacted by endotoxin contamination. EndoToxFree™ DPBS is critical for:
Immunology Research
- Prevents artificial stimulation of macrophages and dendritic cells
- Maintains native immune cell behavior for accurate cytokine profiling
- Essential for LPS-sensitivity studies and inflammation research
Stem Cell Research
- Preserves normal proliferation and differentiation pathways
- Prevents unwanted immune activation that can alter cell fate
- Critical for maintaining stemness characteristics
Drug Discovery & Screening
- Eliminates compound-endotoxin interactions that confound results
- Ensures accurate assessment of drug efficacy and toxicity
- Reduces false positives in high-throughput screening assays
Cell and Gene Therapy R&D
- Maintains native-like cell behavior in vitro
- Essential for accurate translational research
- Supports regulatory compliance for therapeutic development
Endotoxin Level: <0.005 EU/mL (Guaranteed)
Made with Quality Biological’s Water, Cell Culture, WFI Quality
Quality tested for endotoxin level, sterility, and mycoplasma.
For Research or Further Manufacturing Use. Not for Diagnostic or Therapeutic Use.
![]()
Additional information
| Weight | 32 oz |
|---|---|
| Dimensions | 8 × 8 × 8 in |
| Unit Quantity | 500 mL |
| Product Manufacturer | Quality Biological |
Product Documentation
Safety Data Sheet Policy
Tech Brief – EndoToxFree DPBS USP Grade